Page 21 - SAEOPA CLP Guidelines
P. 21
9 GLossARY
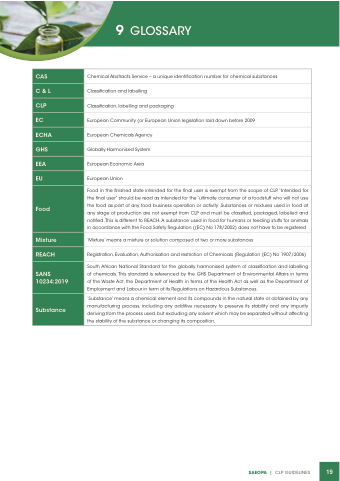
CAS
Chemical Abstracts Service – a unique identification number for chemical substances
C &L
Classification and labelling
CLP
Classification, labelling and packaging
EC
European Community (or European Union legislation laid down before 2009
ECHA
European Chemicals Agency
GHS
Globally Harmonised System
EEA
European Economic Area
EU
European Union
Food
Food in the finished state intended for the final user is exempt from the scope of CLP.“Intended for the final user” should be read as intended for the ‘ultimate consumer of a foodstuff who will not use the food as part of any food business operation or activity’.Substances or mixtures used in food at any stage of production are not exempt from CLP and must be classified, packaged, labelled and notified.This is different to REACH.A substance used in food for humans or feeding stuffs for animals in accordance with the Food Safety Regulation ((EC) No 178/2002) does not have to be registered
Mixture
‘Mixture’ means a mixture or solution composed of two or more substances
REACH
Registration, Evaluation, Authorisation and restriction of Chemicals (Regulation (EC) No 1907/2006)
SANS 10234:2019
South African National Standard for the globally harmonised system of classification and labelling of chemicals. This standard is referenced by the GHS Department of Environmental Affairs in terms of the Waste Act, the Department of Health in terms of the Health Act as well as the Department of Employment and Labour in term of its Regulations on Hazardous Substances.
Substance
‘Substance’ means a chemical element and its compounds in the natural state or obtained by any manufacturing process, including any additive necessary to preserve its stability and any impurity deriving from the process used, but excluding any solvent which may be separated without affecting the stability of the substance or changing its composition.
SAEoPA | CLP GuideLines
19